The bioavailability and toxicity of contaminants such as heavy metal(loid)s is underpinned by their bioaccessibility. Some heavy metal(loid)s are ingested accidentally, such as arsenic (As), cadmium (Cd), mercury (Hg) and lead (Pb). Therefore, gut microbes may play an important role in the absorption of nutrients and heavy metals in the human intestine through their effect on bioaccessibility.
Sources of heavy metal(loid) contamination
Heavy metal(loid) contamination of terrestrial and aquatic environments is mainly a result of human activities – metal mining and smelting, and irresponsible disposal of agricultural and industrial waste are all problematic. Humans encounter the heavy metal(loid) contaminants through water, soil, food and air.
As, Cd, Pb and Hg are some of the most common toxic metal(loid)s that humans ingest accidentally. For example, young children unintentionally ingest Pb-contaminated soil and dust. Pb is also a therapeutic compound in traditional Indian Ayurvedic medicine, used for treating diabetes, diarrhoea and skin diseases. In Bangladesh, deep watertables containing As have contaminated well water and rice (through irrigation), thus creating a pathway for accidental ingestion.
Bioaccessibility of heavy metal(loid)s
The amount of metal(loid) that is absorbed into the blood and systemic circulation (that is, the bioavailable fraction) may vary depending on the:
- nature and solubility of the metal(loid) source – that is, its bioaccessibility
- physicochemical properties of the ingested material.
Bioaccessible metal(loid) concentrations are more important for health and environmental risk assessment than total metal(loid) contents. Therefore, bioaccessibility studies are needed to determine human intake of heavy metal(loid)s from various sources, to accurately assess risk and establish maximum threshold levels for metal(loid)s.
Role of gut microbes
The human microbiome comprises bacteria and other microorganisms that live on and within a human. Most of our 3.8 × 1013 bacteria reside in our gastrointestinal tract, making up the gut microbiome, which:
- provides immune system training and modulation
- digests and ferments energy substrates
- is a source of vitamins and vitamin production
- helps with the structural integrity of the gastrointestinal tract.
These bacteria are essential to normal physiological human functioning. When the gut microbiome is disturbed, it can lead to poor health, including intra- and extra-gastrointestinal disorders.
Ingested contaminants go through the digestive tract, where they will interact with the gut microbes before passing through the intestinal cell wall into systemic circulation. These gut microbes therefore also play an important role in the transformation, bioavailability and, hence, metabolism of heavy metal(loid)s.
Testing the relationship between gut microbes and lead bioaccessibility
Most studies have examined bioaccessibility of heavy metal(loid)s in the absence of gut microbes. In this study, we examined the effect of three gut microbes – Lactobacillus acidophilus, L. rhamnosus and Escherichia coli – from various locations in the gut, on the bioaccessibility of soil-ingested Pb. Bioaccessibility was examined using both Pb-spiked and Pb-contaminated field soil samples from shooting ranges. Pb acetate was used as the reference material (3 samples in total). Bioaccessible Pb was estimated using the in vitro gastrointestinal method, which involves a 2-step sequential extraction: a gastric solution extraction and then an intestinal solution extraction. Both steps were done with and without gut microbes.
The gastric solution extracted more Pb than the intestinal solution for all 3 samples with and without gut bacteria. In the gastric phase (pH 1.5), the solubility of Pb acetate was approximately 100%. When the in vitro solution was modified to the intestinal phase (pH 5.8), Pb acetate solubility decreased to 65%. pH is often one of the main factors that control the solubility of metals, including Pb, in aqueous solutions. In the 3 samples, the absolute Pb bioaccessibility was 45.3–68.7% after gastric phase dissolution. When the solution was modified to reflect intestinal phase conditions, a similar decrease in soluble Pb was seen for the contaminated soils as for Pb acetate. Correspondingly, absolute Pb bioaccessibility decreased to 1.2–11.7%. As Pb dissolution is influenced by parameters such as Pb mineralogy, the source of Pb contamination is likely to significantly influence Pb bioaccessibility.
The difference in Pb bioaccessibility between gastric and intestinal phases can be attributed to the reduced solubility of Pb at pH 5.8. After transitioning from the gastric to the intestinal phase, a significant proportion of solubilised Pb is readsorbed onto soil particles or precipitated at the neutral intestinal phase pH, thereby reducing the bioaccessibility in the intestinal phase. The 2 soil samples showed different bioaccessibility because of the Pb mineral formed in the soil, and the length of interaction between Pb and soil particles.
In our study, gastric and intestinal Pb bioaccessibility decreased in the presence of bacteria, and the effect was more pronounced for gastric bioaccessibility. This may lead to a decrease in Pb bioavailability.
How microbes could modulate lead bioaccessibility
There are several possibilities for how microbes could modulate Pb bioaccessibility. Gut microbes may reduce the bioaccessibility of Pb by immobilising the Pb through adsorption, complexation and precipitation reactions (Figure 1). The microbial cell wall is a natural barrier for metals, including Pb2+. Many microorganisms make extracellular polymeric substances (ESPs) that bind toxic metal cations, thereby protecting metal-sensitive and essential cellular components (Figure 1). The composition of ESPs is very complex, but includes proteins, humic acids, polysaccharides and nucleic acids, which chelate metals with different specificity and affinity. Bacillus firmus, Pseudomonas spp., cyanobacteria, Halomonas spp. and Paenibacillus jamilae are all reported to have ESPs that bind to Pb2+.
Another possibility is through the bonds that metals form with organic compounds. Pb2+ forms strong soluble and insoluble complexes with organic compounds such as tryptone, cysteine, neopeptone, casamino acid and succinic acid. Microorganisms release organic compounds such as short chain fatty acids and carboxylic acids, which are involved in nutrient absorption and energy regulation. When the organic compounds form a complex with metals, the metals are removed from the solution. Lead is also known to react with anions such as chlorides, phosphates and hydroxyl ions to form insoluble precipitates. Several microorganisms precipitate Pb2+ to lower the concentration of free Pb2+ by sequestering it as phosphate salts outside and inside the cell (Figure 1).
Other possibilities for how bacteria affect the bioaccessibility of Pb include the following:
- Some bacteria accumulate Pb2+ in their vacuoles.
- Microorganisms have metallothionein-like proteins that can bind metals, including Pb2+.
- Extracellular enzymes, such as superoxide dismutase excreted by microorganisms, can also bind Pb2+.
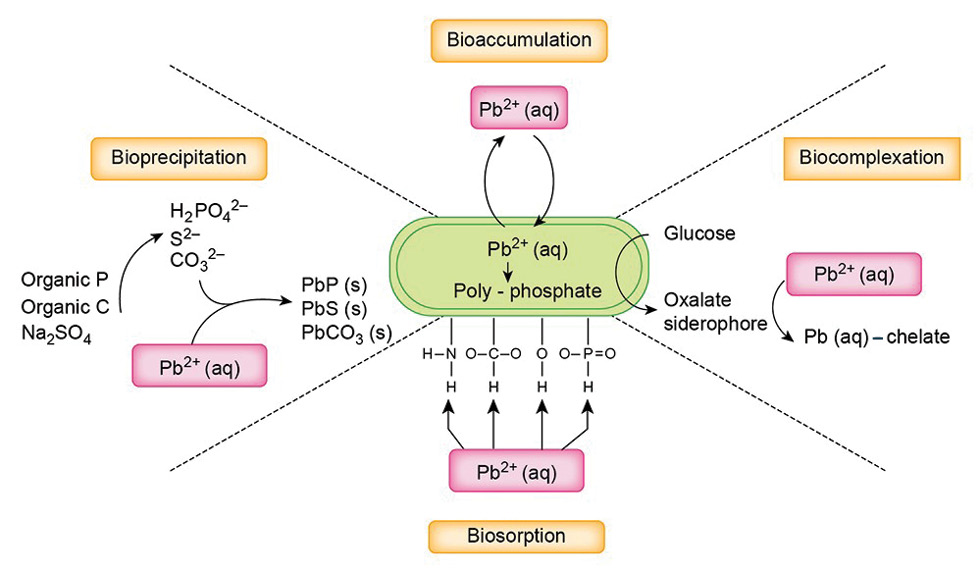
aq = aqueous phase; s = solid phase
Conclusion
Our study showed that gut microbes decreased bioaccessibility of Pb, which is likely to affect its bioavailability, intestinal absorption and toxicity. These microbes can act as the ‘guardians of the gut galaxy’. The effect of gut microbes on bioaccessibility may be attributed to the bioimmobilisation of Pb through adsorption, precipitation and complexation reactions. Therefore, bioaccessibility measurements must be done in the presence of gut microbes, especially for ingested contaminants.
It is important to point out that the human gut hosts many microbial species, including bacteria, fungi and archea. We looked at the effect of only 3 bacteria on the bioaccessibility of Pb. Future studies should focus on the effect of composite gut microbial culture on bioaccessibility and subsequent bioavailability of toxic metal(loid)s.